|
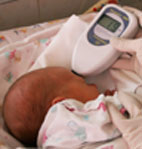
Commonly in newborns bilirubin determination
is performed on a forehead near nose or on the upper part
of the breast. Only a slight touch of a mobile measuring
head to a baby’s derma is needed for measuring. The
result is given in international units for transcutaneous
bilirubin index (TBI). This index has a high correlation
degree with plasma bilirubin concentration (0,92).
Power is supplied by 3 AA batteries, the lifetime of a
single battery set exceeds 100,000 measuring cycles. No
turning on/of is needed as BILITEST is continuously in
standby power-saving mode. Measuring cycle takes no more
than 1-2 sec. The cycle starts automatically on touching
baby's derma with a sensitive mobile head. Next measures
can be done in each 5 seconds with automatic erasing of
the previous readings. Measurement range is 2-50 units
TBI (20-500 micromol/l), photometric accuracy - 10%.
Dimensions - 65x135x35 mm, weight - 150 gr.
|
|


